|
(Recall that the number of valence electrons is indicated by the position of the element in the periodic table. While inner electrons (those not in the valence shell). Add together the valence electrons from each atom. Valence electrons are the highest energy electrons in an atom and are therefore the most reactive. Since carbon has 6 electrons in total, it will have 4 valence electrons as shown below: Fig. All neutral elements will have the same number of protons and electrons, which is equal to the atomic number. Do you notice any patterns in the number of valence electrons The number of valence electrons increases going left to right across a period. Carbon is the 6th element in the periodic table. You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. Determine the total number of valence electrons in the molecule or ion. Valence electrons on the periodic table Bohr models for the first three periods of the periodic table are shown below. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). 
Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. The periodic table is arranged by atomic weight and valence electrons. Thus, it is convenient to separate electrons into two groups. The path a specific element will take depends on where the electrons are in the atom and how many there are. 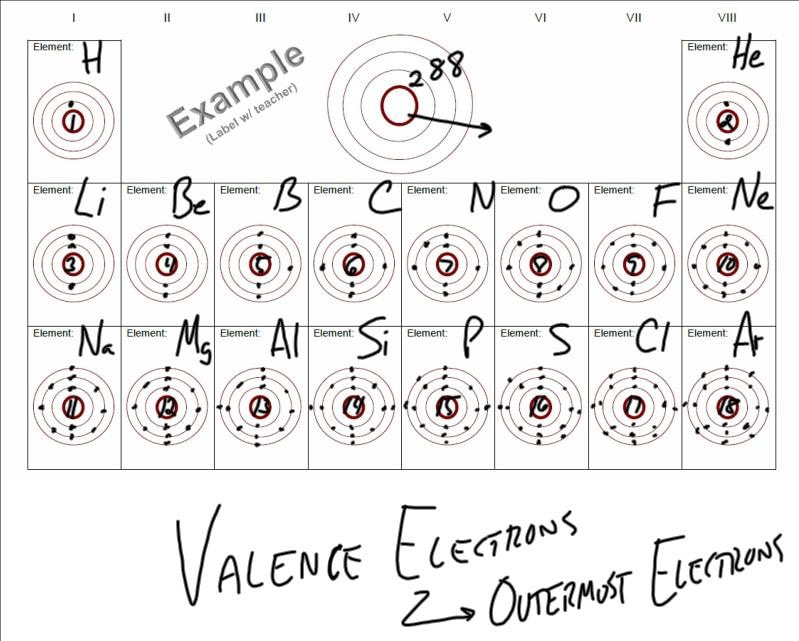 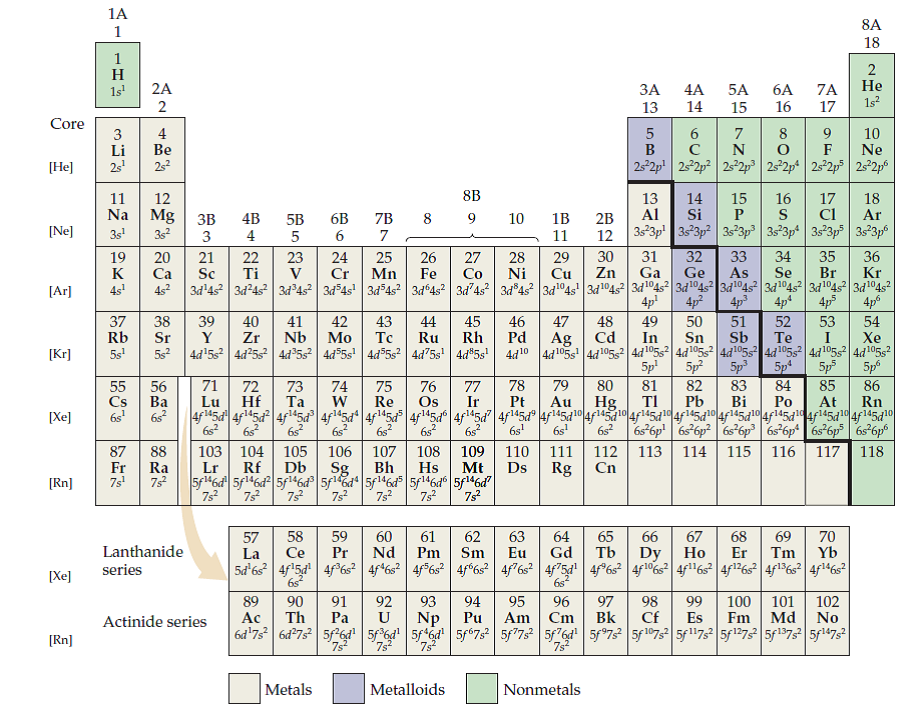
\,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms. The position of an atom in the periodic table represents the number of valence electrons present in the outermost shell of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
